Covishield Side Effects: Doctors’ Group Urges Govt to Review all Covid Vaccines
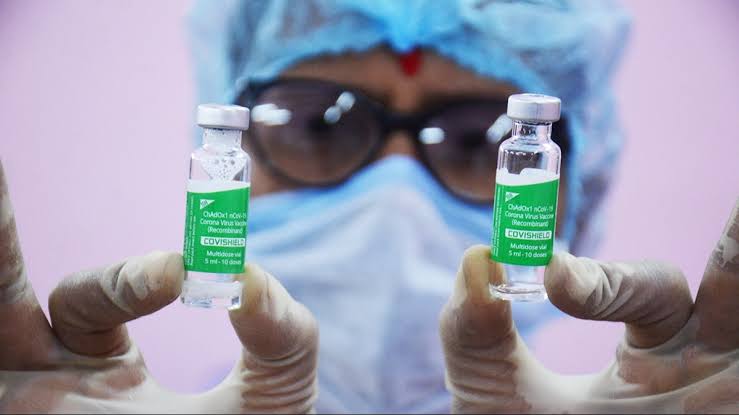
A group of doctors, organized under the Awaken India Movement, voiced concerns about the safety of the Covishield vaccine manufactured by the Serum Institute of India. This came in the wake of AstraZeneca’s acknowledgment of rare blood clot side effects in its COVID-19 vaccine.
Dr. Tarun Kothari stressed the need for a comprehensive review of all COVID-19 vaccines’ scientific basis. He also called for the implementation of active surveillance systems to detect adverse events promptly.
Dr. Sujata Mittal highlighted the lack of complete information on vaccine side effects prior to their administration. She emphasized the low awareness surrounding vaccine injuries, particularly in India.
Dr. Mittal also mentioned the issue of menstrual cycle abnormalities reported by numerous women post-vaccination, later confirmed as a side effect in a study published in September 2022.
The AIM has been documenting COVID vaccine-related deaths in India since 2021, urging authorities to investigate. They also demanded compensation for victims and their families, along with the establishment of fast-track courts for justice.
AstraZeneca’s global withdrawal of its COVID-19 vaccine, known as Covishield in India, was mentioned. The Serum Institute of India stopped manufacturing additional doses in December 2021, citing rare side effects, including thrombosis with thrombocytopenia syndrome (TTS).
The withdrawal was initiated due to an excess of updated vaccines available since the pandemic’s onset. The company had disclosed rare side effects, including TTS, in packaging inserts in 2021.





